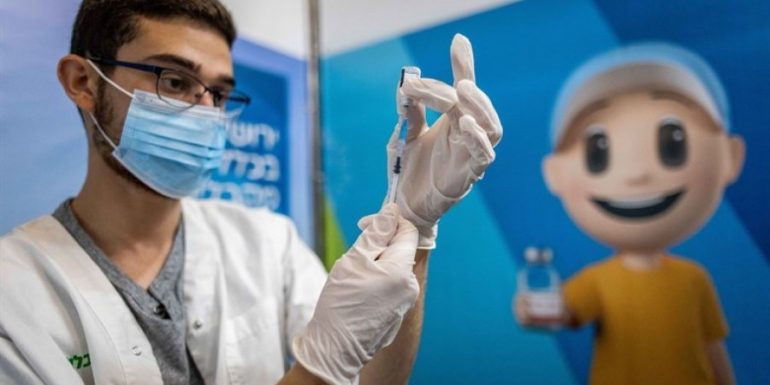
Pfizer and BioNTech announced Tuesday that they are seeking approval from the US Food and Drug Administration (FDA) to vaccinate children up to 6 months of age against Covid-19.
If approved, children aged six months to five years can be vaccinated against it Covid-19 with the Pfizer and BioNTech vaccine. Currently, children five years of age and older can be vaccinated in the United States with the Pfizer vaccine - but not with Moderna and Janssen, which are limited to people over 18 years of age.
Pfizer claimed in a press release that there was an "urgent public health need" for children under the age of five to be vaccinated against coronavirus.
"This application is for the approval of the first two doses of 3 μg of a scheduled initial three-dose series in this age group," Pfizer explained, adding that the company is still awaiting trial data for a third dose for young children.
The cases COVID-19 "and the relative hospitalization of children increased dramatically in the United States during the rise of the Omicron variant," the press release said, noting that "children under 4 accounted for 3,2% of total hospitalizations" several weeks ago.
Pfizer also notes that, if approved, its vaccine will be the first available for children under five in the United States. According to the CDC, more than 80% of Americans age five and older have received at least one dose of the vaccine Covid-19. About 67,9% of Americans over the age of five are "fully vaccinated."
Source: in.gr